Original Articles: 2022 Vol: 14 Issue: 3
The effect of some Medicinal and Aromatic Plant Extracts on Rhizopus Stolonifer in Central Darfur State
Mohammed TMS1*, Saaeed Dehiash AEB2
1Department of Horticulture, University of Zalingei, Zalingei, Central Darfur, Sudan
2Department of Biology, University of Zalingei, Zalingei, Central Darfur, Sudan
- Corresponding Author:
- Mohammed TMS
Department of Horticulture, - University of Zalingei,
Zalingei,
Central Darfur,
Sudan
Received: 07-Mar-2022, Manuscript No. JOCPR-22-51825; Editor assigned: 09-Mar-2022, PreQC No. JOCPR-22-51825 (PQ); Reviewed: 23-Mar-2022, QC No. JOCPR-22-51825; Revised: 28-Mar-2022, Manuscript No. JOCPR-22-51825 (R); Published: 07-Apr-2022, DOI:10.37532/ 0975-7384.2022.14(3).017
Abstract
The need for natural antimicrobials is paramount to avoid harmful synthetic chemicals. This study investigated the antifungal activity of some medicinal and Aromatic Plants against mold. Rhizopus stolonifer is one of the strong parasitic pathogens, which can cause fruits and vegetables causing economic losses. The study aimed to determine the antifungal activity by using the natural compounds of (Clove, Neem, Tea, and pomegranate.). Different concentrations (0.1, 0.01, 0.02, and 0.05 ppm) prepared from these extracts inhibited the growth of the test pathogens and the effect gradually increased with concentration. Clove aqueous extracts showed a broad antifungal spectrum at all studied concentrations, followed by Tea, Neem, and Pomegranate. The study suggested that Clove was to be significantly very effective against Rhizopus stolonifer. Result proves that pomegranate was not very effective in controlling the mold in this study in the mean of a few toxic compounds to Rhizopus spp.
Keywords
Antifungal; Rhizopus stolonifer; Clove; Neem; Tea; pomegranate
Introduction
Rhizopus stolonifer is a strong parasitic pathogen, which can cause postharvest rot of peach, strawberry, grape, and other fruits and vegetables [1,2]. The fungus mainly penetrates the host body through external scratches during harvesting period, transportation, and sale. The mycelial mass grows on the fruit surface and produces long mycelial stolons. Growth, reproduction, and infection rate are very fast, and the spores can spread with airflow, once it
invades the host, it will spread rapidly, and cause decay and softness within 1-2 days, which seriously affects the transportation and sales of fruits and vegetables [3]. Researchers suggested that the use of multiple microbial strains in combination with R. nigricans as a starter culture could be a novel and effective approach to improve the nutrition and safety of fermented soybean food products of doenjang [4].
Rhizopus species have an especially vital role in the food industry due to their ability to produce thermostable poly galacturonases, which hydrolyses α-1, glycosidic bonds between galacturonic acid residues and thus affect the plant
cell wall [4]. In particular, R. oligosporus is used to ferment soybeans to make tempeh, which has many health benefits including potent anti-radical activity [4].
Biopesticides were classified into different categories, such as microbial pesticides, plant-incorporated protectants, and biochemical. Biopesticides are a crucial component of integrated pest management programs for pest control, which lead to more natural alternatives to chemical pesticides that are eco-friendly and safer [5].
The application of fungicides is one of the easily available and adopted methods in plant disease management.
However, regular and indiscriminate use of fungicides results in many adverse effects on human health as well as on the environment. However, due to increased awareness about the risk involved in the use of fungicides like residual
problems, the use of other alternative methods in the management of plant diseases is gaining importance [6].
Many publications have featured botanical pesticides with skewed interest towards the management of insect pests. This review brings together information regarding botanical pesticides, their phytochemical composition, and mechanisms of action against pests of importance in agricultural production [7].
Integrated pest management is a dynamic approach in which pesticides are essential tools for pest suppression. Nevertheless, pest management tools should be cost-effective and present minimal risk to humans and desirable
components of the environment, which provides an opening for bio-pesticide products [8].
Biopesticides have several advantages over their chemical counterparts and are expected to occupy a large share of the market in the coming period [9].
Neem has been used for a long time in agriculture and alternative medicine. It is considered as a safe medicinal plant
having numerous biological properties without any adverse effect [10]. On fungal isolates from yam, the mean percentage zones of inhibition of all extracts of the neem plant parts showed more effectiveness than the positive
control (Keto-conazole) against L. theobromae growth. Aqueous stem bark extract with 93.80 ± 1.78% zone of inhibition showed comparable effectiveness to the control (100% inhibition) against A. ochareus growth, while against S. rolfii growth, there was good inhibition by all the extracts though not to the extent as that of the control [11].
Xie, et al. found that the inhibitory effect of clove, geranium, agal wood and lavage growth of R. stolonifer were the
best, and the inhibition rate was 100%, which were significantly higher than other essential oils except for ilex [12].
The application of clove buds as a botanical pesticide for future use against nematodes is highly promising since clove is the sixth major plant grown on Bangka Island, and the market value of clove has decreased sharply over the
last years [13].
Even though it is, true that under field conditions other biotic or abiotic factors may influence the expression of the
antagonism, the in vitro assays allow the selection of Trichoderma species suitable as biocontrol agents, but further research is needed to validate the most prominent strains under systems that resemble natural conditions or under field conditions. It may conclude that it is possible to isolate antagonistic fungi from several soils with different use such as forestry and agriculture. In addition, Trichoderma strains are appropriate agents for controlling the growth and sclerotia of either Ss or Sm [14].
Assays were conducted to evaluate the growth of R. stolonifer on glucose or cutin as a sole carbon source. The
pathogen grew on both media and higher esterase activity observed in the cutin medium. Wounded and unwounded peaches and nectarines were inoculated with R. stolonifer spore suspension in water or modified Van Etten nutrient solution 1.
In addition, evolutionary changes in both hosts and natural enemies might have unexpected consequences on levels of bio-control exerted by enemies [15].
High in vivo inhibitory effects of M. spicata and C. martinii EOs were observed at 10% concentration, with 100 and 78% reduction, respectively, in the R. stolonifer induced spoilage [16].
Growth inhibition was highest in Curvularia sp. which did not grow beyond the initial point of its radial growth before the introduction of the extract, while the lowest effect was observed in Rhizopus sp [17].
Researchers often study the antagonism between microbes to produce novel, effective antimicrobials but stop short of the large compounds secreted into the nearby environment. It found that bacteria produce antifungals that can volatilize and inhibit fungal growth from a distance [18].
The resulting film was homogenous, flexible, luminous, and slightly dark, with cumulative viscosity which means that total viscosity is the result of each component viscosity. The CS-OM film demonstrated a strong antifungal
effect against Rhizopus stolonifer in vitro and situ conditions and increased the shelf life of tomatoes [19].
Extracts of various parts of neem, but especially of the seeds, has been shown to possess feeding detergency, repellency, toxicity, and growth disruptive properties to numerous species and stages of insects of many orders, and this has earned for the tree its reputation as the "wonder tree". A comprehensive review of the entomological properties of neem has been published [20]. A scientific study explained that the antibacterial effect of phenolic compounds of pomegranate could be increasing with the presence of organic acids [21]. Herein, the biochemical composition of the Pomegranate Peel Extract (PPE), its efficacy in food preservation, and antimicrobial activities are
discussed to provide a comprehensive guide for farmers, food processing, and storage sectors, and academia [22-23].
In vitro, antifungal activity test showed high growth inhibition at the concentration (4 %) of each plant (extract), and suggest using plant extracts as natural treatments for increasing storability and quality of Jerusalem artichoke fresh tubers and inhibition of fungi diseases [24].
Materials and Methods
Plant Materials
All plants materials Tea (Camellia sinensis (L.) Kuntze); Neem (Azadirachta indica); Pomegranate (Punica granatum); clove (Syzygium aromaticum) used in the experiment were be purchased from Zalingei spices market. Sabouraud was been brought from the laboratory of biology, Department of Biology, Faculty of Education. 46.0 g powdery Potato Dextrose Agar (PDA) medium was weighed and added to 1000 mL distilled water, autoclaved at
115℃ for 20 mins, and then stored at 4℃. Rhizopus stolonifer was isolated from the infected bread.
Isolation of Rhizopus sp.: Bread mold ripped and placed on Sabouraud Dextrose Agar (SDA) and incubated for five days. Rhizopus sp. was been purified in a new medium.
Purification of Rhizopus sp.: Purification of Rhizopus sp. was been conducted by eliminating ecto-symbiont bacteria using three methods. The three methods were been modified ring method, simple method, and elimination method using ciprofloxacin antibiotics.
Glass ring with 15-20 mm in diameter, 1-2 mm wall thickness, and 8-10 mm height was passed into the fire briefly and cooled again, then pressed in the middle of the SDA as deep as about 5 mm, in which the existing heat residue
in the glass ring would be embedded in agar. One needle of fungus was been inoculated into the ring, and then its growth was observed for one to two days until the initial hyphal growth. The hyphae were transferred to new sterile SDA media. After five days of incubation at room temperature, the isolate was been purifying by cutting a small amount of mycelium and placed on a sterile petri dish. A rectangular SDA was been placed on the mycelium, and then small pieces of SDA were placed above the rectangular piece. The culture was incubated until the fungus grew
through a rectangular agar and small pieces agar above it. Small fragments of agar will be transferred to the new
sterile SDA and incubated. Three days culture inoculated into Trypticase Soy Broth (TSB) containing 0.05 mg mL-1
ciprofloxacin; then incubated overnight at 30°C. After incubation, the broth medium was been centrifuged at 4000 rpm for five minutes. The filtrate and supernatant were inoculated separately in SDA-containing antibiotics. It was been repeated three times.
Results and Discussion
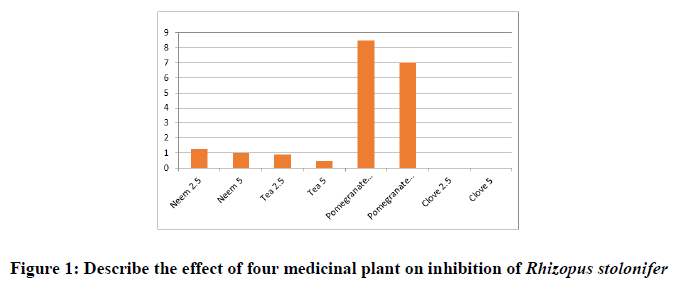
Data presented in shape [1] showed the variation of inhibition among the four plant aqueous extracts (water extract) tested against Rhizopus stolonifer. Clove aqueous extract expressed the best result in this treatment and prevent mycelia growth in all concentrations. While the aqueous extract of Tea (Camellia sinensis (L.) Kuntze); better than Neem (Azadirachta indica); and followed by Pomegranate (Punica granatum) (Figure 1).
Clove was more effective than all tested plant extract to inhibit microorganisms, which might be a result of its
higher eugenol concentration [25]. Similar observations were found with effective in inhibiting the growth of Aspergillus niger by clove (Syzygium aromaticum), even at lower concentrations [26]. Several scientists reported the
inhibitory effect of clove oil on growth and aflatoxin production by A. flavus, in maize, clove oil effectively fought
against aflatoxin formation by A. flavus for 10 days [27]. Similar observation demonstrated that clove oil could be a
promising natural fungicide for an effective bio-control, non-toxic bio-preservative, and an eco-friendly alternative to synthetic additives against A. flavus in Georgia peanuts [28].
The modes of neem action include repellence, inhibition, denaturation of proteins, and other effects depending on the type of botanical compound and pest. For instance, pesticides from the pyrethrum target the nerve cells of insects leading to paralysis and later died while neem-based pesticides have anti-feedant and repellence properties, induce molting abnormalities, hinder oviposition and disrupt the endocrine system [7]. This research has shown that the potential use of crude and aqueous extracts of Azadirachta indica Leaves for controlling fungal pathogens was due
to the phytochemicals present in the extract and the presence of these bioactive compounds in the Azadirachta indica Leaf extract is thought to be responsible for the antifungal activity [29].
Various neem plant parts extracts showed inhibition against fungal growth from isolates of rotted cocoyam [11]. In scientific research, it was observed that the two molds (Fusarium exosporium and Alternaria spp.)species have been controlled by the alcoholic extract of the leaves and stems of Proboscidea parviflora [30].
Moriniafungin showed protective control efficacies against Rhizopus rot on apple and peach fruits [31]. In vitro
microbiological analyses were been used to evaluate the presence of the microorganisms after fruit immersion in alkaline solutions, while the disc diffusion method was used to study the inhibition of microorganism growth. According to the results, alkaline solutions at pH 13 can be utilized to control Botrytis cinerea and Rhizopus stolonifer in strawberries [32]. Preventive field fungicide sprays control Rhizopus soft rot reducing field inoculum levels, fungicide sprays also reduce the incidence of fruit lesions, caused by other fungi since Rhizopus can act as courts of entry into the papaya fruit [33].
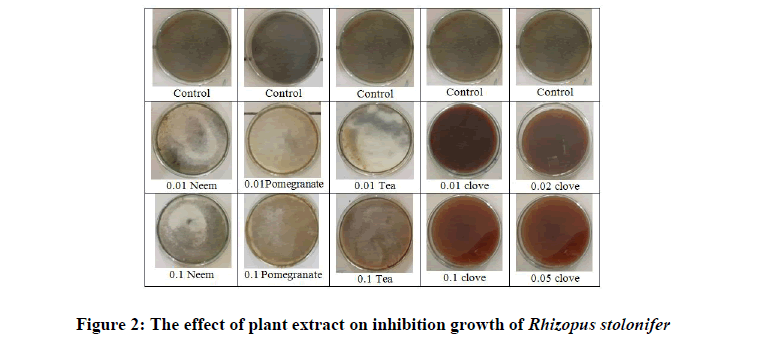
Pomegranate (Punica grantum) was found to have microbiological activities that might be to the high phenolic and punicalagin content, which significantly correlated with antifungal activity [23]. The effect of aqueous pomegranate extracts was of a similar pattern like methanolic extracts; wherein the following order of decreasing antifungal effect was observed for tested fungi: (A. niger; P. citrinum; R. oryzae; T. reesei, and M. indicus) (Figure 2) [34].
The unique set of secondary metabolites produced by plants may play an important role in sustainable pest
management as new products directly, as novel chemical frameworks for synthesis and/or for identifying original modes of action. The potential of plants and their secondary metabolites for plant health could be used in different strategies: employing the whole plant, crop residues, and part of plants, and using plant chemicals and extracts in integrated or ecological pest management acting directly on the target pest or inducing resistance [35].
In a study, it was demonstrated that green tea was found to be very effective on C. albicans, Streptococcus mutans,
and Lactobacilli spp, which might be resulted in the presence of phenols compound and quinines [36].
Several studies have shown that the antimicrobial property of Tea (Camellia sinensis) is due to the presence of polyphenols. Specific antioxidant polyphenols called catechins play an important role in green tea’s inhibition of microbial growth. Anti-microbial activities of tea extracts are very selective [37,38].
Conclusion
From the results of the study, it concluded that Clove was to be significantly effective against Rhizopus stolonifer.
The very low concentration (i.e., 0.1 ppm) of clove could cause 100% inhibition of growth and its (0.01; 0.02; 0.05)
concentration proved fungicidal. However, further extensive in vitro and in vivo studies should be undertaken on
whether these extracts can be used in the treatment of Rhizopus stolonifer infections or remedy.
References
- Baggio JS, Gonçalves FP, Lourenço SA, et al. Plant Pathol. 2016;65(4):633-642.
- Hernández-Lauzardo AN, Bautista-Baños S, Valle MGV. Rev Mex Fitopatol. 2006;24(1):65-69.
- Niurka A, Lauzardo H, Bautista S, et al. Rev Mex Fitopatol. 2006;24(1):65-69.
- Shukla S, Bahuguna A, Park HK, et al. Sci Rep. 2020;10(1):1-10.
- Samada LH, Tambunan USF. Online J Biol Sci. 2020;20(2):66-76.
- Symposium N. Souvenir & abstracts. 2021.
- Lengai GMW, Muthomi JW, Mbega ER. Sci African. 2020;7:e00239.
- Falahzadah MH, Karimi J, Gaugler R. Egypt J Biol Pest Control. 2020;30(1).
- Kumar J, Ramlal A, Mallick D, Mishra V. Plants. 2021;10(6):1-15.
- Road LB. 2020.
- Ezeonu CS, Imo C, Agwaranze DI, et al. Chem Biol Technol. 2018;5(1):1-9.
- Xie Y, Jiang L, Li J, et al. IOP Conf Ser Earth Environ Sci. 2021;792(1):012014.
- Taniwiryono WD, Berg H, Riksen J, et al. Open Nat Prod J. 2009;2:77-85.
- Medina IV, Cerrato FR, Alarcón A, et al. Rev Mex Micol. 2010;31:53-63.
- Thomson LJ, Macfadyen S, Hoffmann AA. Biol Control. 2010;52(3):296-306.
- Filho JGO, Silva G, Egea MB, et al. Biointerface Res Appl Chem. 2021;11(5):13244-13251.
- Olufemi AA, Joseph OAT, Grace FA. 2014;3(1):106-109.
- Stajich JE, Borneman J, Stajich JE, et al. 2019.
- Hernández ODR, Sánchez AÁ, López MR, et al. TIP Rev Espec en Ciencias Químico-Biológicas. 2019;22:1-9.
- Jacobson M. Botanical Pesticides. 1989:1-10.
[Crossref]
- Orak HH, Demirci AŞ, Gümüş T. Electron J Environ Agric Food Chem. 2011;10(3):1958-1969.
- Chen J, Liao C, Ouyang X, et al. J Food Qual. 2020.
- Rongai D, Pulcini P, Di Lernia G, et al. Horticulturae. 2019;5(3):1-9.
- El-Awady A, Ghoneem K. J Plant Prod. 2011;2(12):1815-1831.
- Mutlu-Ingok A, Devecioglu D, Dikmetas DN, et al. Molecules. 2020;25(20).
- Akromah ZN, Achmad, Jayanegara A. IOP Conf Ser Earth Environ Sci. 2021;667(1).
- Habschied K, Krstanović V, Zdunić Z, et al. Fungi. 2021;7(5).
- Achar PN, Quyen P, Adukwu EC, et al. J Fungi (Basel). 2020;6(4):383.
- Keta JN, Suberu HA, Shehu K, et al. Sci World J. 2019;14(1):98-102.
- Sharief TM, Albala MMA. Int J Plant Sci. Ecol. 2015;1(2):22-25.
- Park MY, Park SJ, Kim JJ, et al. Plant Pathol J. 2020;36(6):570-578.
- Santacruz S, Cedeño J. Rev Fac Nac Agron Medellin. 2021;74(2):9615-9619.
- Salman MA. Doctoral dissertation. 2005.
- Soeda M, M Otomo, M Ome, et al. Nippon saikingaku zasshi Japanese J Bacteriol. 1966;21(10):609-614.
- Pino O, Sánchez Y, Rojas MM. Revista de Protección Vegetal. 2013;28(2):81-94.
- Hanci H, Coşkun MV, Uyanik MH, et al. Bezmialem Sci. 2019;7(2):107-112.
- Se C, Muturi M, Bii C. J Med Plant Res. 2015;9(42):1070-1080.
- Surapuram V, Setzer WN, McFeeters RL, et al. Nat Prod Commun. 2014;9(11):1603-1605.