Original Articles: 2025 Vol: 17 Issue: 1
Effect of Terminal Methoxyethyl Tail on Homologous with Imine and Ester Groups on Mesomorphism
Chitra A Padh1*, Mikita Shah1, Patel HN2, Baria GN3, Tarun M. Patel3
1Department of Chemistry and Chemical Sciences, Sabarmati University, Ahmedabad, India
2Department of Science, Shree Maneklal M. Patel Institute of Science and Research, Kadi Sarva Vishwavidyalaya, Gandhinagar, India
3Department of Chemistry, Sir P.T. Science College, Modasa, India
*Corresponding Author:
Received: 15-Oct-2024, Manuscript No. JOCPR-24-150182; Editor assigned: 18-Oct-2024, PreQC No. JOCPR-24-150182 (PQ); Reviewed: 01-Nov-2024, QC No. JOCPR-24-150182; Revised: 15-Jan-2025, Manuscript No. JOCPR-24-150182 (R); Published: 22-Jan-2025, DOI:10.37532/0975-7384.2025.17(1).242.
Citation: Chitra AP, et al. 2025. Effect of Terminal Methoxyethyl Tail on Homologous with Imine and Ester Groups on Mesomorphism. J. Chem Pharm. Res., 17:242.
Abstract
A new type of liquid crystals with p-methoxyethyl end chains was prepared: p-methoxyethyl 4-(4- alkoxybenzylideneamino)benzoate. All twelve compounds synthesized in the series show mesomorphism. Methoxy to nhexadecyloxy derivatives exhibit enantiomeric smectic A mesophase. Nematic mesophase starts with nmethoxy derivative as enantiomer and continues until nbutoxy homologue is synthesized. Analysis was carried out on Optical Measuring Machine (POM). Some derivatives detected using Differential Scanning Calorimetry (DSC) techniques were used to obtain thermograms of phase change, enthalpy and entropy change. Some homologues were characterized using infrared spectroscopic methods, proton magnetic resonance and ultraviolet spectroscopy.
Keywords
Methoxyethyl tail; Schiff base; Smectic A; Nematic; Mesogenic
Introduction
Mesomorphism in liquid crystals is accompanied by significant changes in molecular structure, partial or complete orientation, and intermolecular/intramolecular interactions [1]. The last two depend on the molecular structure itself [2. Therefore, small changes in the geometry of a molecule can cause significant changes in its phase transitions [3]. This change in geometry can be a change in the alkyl/alkoxy substituted (OR) chain, a change in the unsymmetrical core, a change in the linking group, and finally a change in the group [4]. The size and length of the terminal and linking groups, as well as the orientation of their linkages, play an important role in the phase transition, while the alkyl or alkoxy (OR) chains provide the molecular orientation required for the step generation [5]. Among all the main parts of liquid crystal molecules, the terminal group is an important group [6]. The saturation of the terminal chains plays an important role in the thermal and electrooptic properties of the liquid crystal, where the order of the [C=C] double bond is related to the properties of the liquid crystal [7]. Also many liquid crystals using small cyano [-NO2] or halogen have been used or chain breaking has been found to lower the melting point of the alkyl and remove it [8].
Come up with a long chain. Liquid crystal derivatives containing chiral (menthol and lactic acid combined with various bond groups are seen to exhibit "De Vries" type behavior in short molecules, exhibiting SmA*-SmC*. The transition phase is lower. Prajapati and Vora have made extensive studies on alkoxy chain broken liquid crystals having low melting points [9]. By combining different LC molecules with nonlinear linking groups and incorporating alkoxy end chains, unconventional molecular structures with conformational terminal arrangements have been created [10]. The presence and position of the linking group influences the overall polarity of the molecule [11]. Materials with imine (CH=N) linking groups can exhibit photochromic, thermochromic, and piezochromic behavior and also exhibit some adverse optical properties [12]. They also have potential applications in highspeed materials, liquid crystals, passive devices, molecular switches, and polymer actuators [13]. Prajapati and Patel reported the synthesis of Schiff base esters with chloroethyl, methoxy, ethoxy, and butoxy tails and investigated the effect of various polar groups on the terminals [14]. Ahmed H. et al. developed and studied the naphthyl base and studied the effect of naphthyl overhang on the stability of the mesophase. Ahmed H. et al [15].
Literature Review
Biphenyl liquid crystals with end groups/chains and linking groups have been created together, where the effect of methylene and ether linking groups on the curvature of the molecule only produces linear progenitors, eventually affecting the chain length TNTBN to a smaller extent than TNI [16]. Similarly, the liquid crystal with biphenyl core and ester bond with (S)-2-methylbutylactic acid group at one end and alkoxy chain and chlorine substituted son molecular daughter at the other end, when compared with the following, it is shown that the addition of two lactate groups at the ends reduces TiSO compared to the compound with an alkyl chain attached to oxygen at the other end, and after modification next to it this is further improved by the addition of lactone molecules which increases the length [17]. The length of the molecule eliminates any crystallinity and therefore the crystalline liquid becomes poor when the length of the molecule is increased beyond a point [18]. Also to understand the effect of the end group, Zhu et al (Tables 1 and 2). The aim of this synthesis is to investigate the effect of chain length and shape on the mesogenic properties of liquid crystals [19].
|
Compound no. |
R=-CnH2n+1 |
Cr |
|
SmA |
|
N |
|
I |
|
1 |
1 |
• |
166 |
• |
-174 |
• |
230 |
• |
|
2 |
2 |
• |
162 |
• |
175 |
• |
202 |
• |
|
3 |
3 |
• |
150 |
• |
191 |
• |
226 |
• |
|
4 |
4 |
• |
153 |
• |
201 |
• |
221 |
• |
|
5 |
5 |
• |
155 |
• |
|
|
216 |
• |
|
6 |
6 |
• |
141 |
• |
|
|
212 |
• |
|
7 |
7 |
• |
142 |
• |
|
|
210 |
• |
|
8 |
8 |
• |
131 |
• |
|
|
206 |
• |
|
9 |
10 |
• |
101 |
• |
|
|
239 |
• |
|
10 |
12 |
• |
134 |
• |
|
|
221 |
• |
|
11 |
14 |
• |
141 |
• |
|
|
201 |
• |
|
12 |
16 |
• |
145 |
• |
|
|
186 |
• |
|
Note: ()=monotropic value; Cr=crystalline solid; SmA=Smectic A phase; N=Nematic phase; I=Isotropic liquid phase; •=phase exists. |
||||||||
Table 1: Transition temperatures (ºC) of the series I compound.
| Sr. no. | Molecular formula | %Theoretically | %Observation | ||||
| C | H | N | C | H | N | ||
| 1 | C32H37O6N | 72.31 | 6.96 | 2.63 | 72.11 | 6.99 | 2.43 |
| 2 | C34H40O6N | 73.11 | 7.16 | 2.5 | 73.21 | 7.26 | 2.58 |
| 3 | C36H43O6N | 73.84 | 7.35 | 2.39 | 73.81 | 7.25 | 2.29 |
Table 2: Elemental analysis of octyloxy, decyloxyanddodecyloxy derivatives.
Synthesis
Information now required to make a homologous compound: Phydroxybenzaldehyde, appropriate n-alkyl halide, anhydrous potassium carbonate, anhydrous acetone, p-aminobenzoic acid, glacial acetic acid, absolute ethanol, 2-methyl Oxyethanol, sulfuric acid. N-dicyclohexylcarbodiimide (DCC), 4dimethylaminopyridine (DMAP) and Tetrahydrofuran (THF) were used in this work. The solvent was distilled and dried before use [20]. Fourier transform infrared spectra were recorded on a Shimadzu IR408 spectrometer using KBr particles possible. Polarized optical microscopy shows the phase change as the phase is heated. Shimadzu DSC60 (Differential Scanning Calorimeter) measures the phase change between cooling and heating at 10°C min-1. N-Alkoxybenzaldehyde was synthesized from 4-hydroxybenzaldehyde using the method of Vyas and Shah. The total yield of 4-alkoxybenzaldehyde was 62% to64%. Alkoxybenzylideneamino)benzoic acid was produced by refluxing 4-alkoxybenzaldehyde and 4-aminobenzoicacid in the presence of ethanol and glacial acetic acid for 34 h. The product was filtered and purified by columnchromatography and confirmed by 1H NMR and IR analyses. Synthesis of β-methoxyethy-l-4-hydroxybenzoate Theesterification of 4-hydroxybenzoic acid and 2-methoxyethanol is as described in the scheme. Product size of β-methoxyethyl-hydroxybenzoate. The ester was distilled off under reduced pressure. Melting point: 87°C (reportmelting point: 87°C); overall yield of p-methoxyethyl-4-hydroxybenzoate is 80%.
Discussion
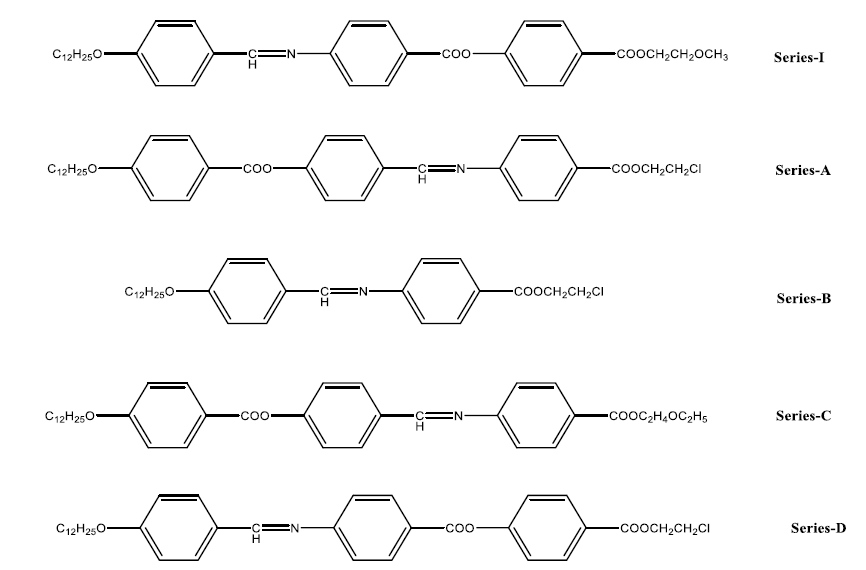
4-((2-methoxyethoxy)carbonyl)phenyl(E)-4-(((4-n-Alkoxybenzylidene)amino)benzoate) synthesis of 0.1 mol 4-(((4-n-Alkoxybenzylidene)amino)benzoic acid, 0.1 mol p-methoxyethyl-4 hydroxybenzoate, 0.1 mol DCC and 0.1 mol DMAP were stirred in anhydrous THF at room temperature overnight and the insoluble solids were removed by filtration through ethanol. synthesized using the same method. The yield is usually 60,65%. The graph of heat transfer is prepared as shown in Figure 1. It produces many results and hence we can determine the nature of the different stages occurring during the transfer. In this study the enthalpy of the derivatives of series I (n=10) was measured by differential scanning calorimetry studies. The readings are summarized in Table 3. Compare the previous results. Micrograph texture of the focal conical structure SmA mesophase (series I; n=10) when cooled at 240°C. The droplet nematic mesophase (series I; n=10) when cooled at 205°C. Comparison with other series A, B, C and D of the samples in Table 4 shows that the smectic mesophase of series I decreases by 8°C. It can be stated that the temperature change of series I is generally 10°C higher than series A. While there is an Azomethine (CH=N) group in the central bond of series I, there is an Ester (-COO) bond in the same position in series A, so as the temperature change in the mesophase increases, series I becomes less. Series A is more polarizing. (COO) Contact us. The components of series I are longer than the components of series B due to an additional aromatic ring and a central ester (-COO-) bond. Gray describes the increase in molecular length and the effect of polarizability anisotropy. It can also be noted that the transition temperature of series I is approximately 117°C higher than that of series C. Series I has a Methoxy (OCH3) end group, while series C has an ethoxy end group. The presence of polar terminal Methoxy (OCH3) group increases the overall polarizability of series I compared to series C, which makes series I have a higher temperature and a larger mesophase thermal range with Azomethine (CH=N) group. Series C has an Ester (COO) bond at the same position in the middle bond, hence series I is more stable than series C due to more mesophase and polarizability temperature changes. It is also seen that the transition temperature of smectic phase of series I is 17°C lower than series D, respectively. Series I has Methoxy (-OCH3) end groups, while series D has more polar Chlorine (-Cl) end groups (Figures 1-10).
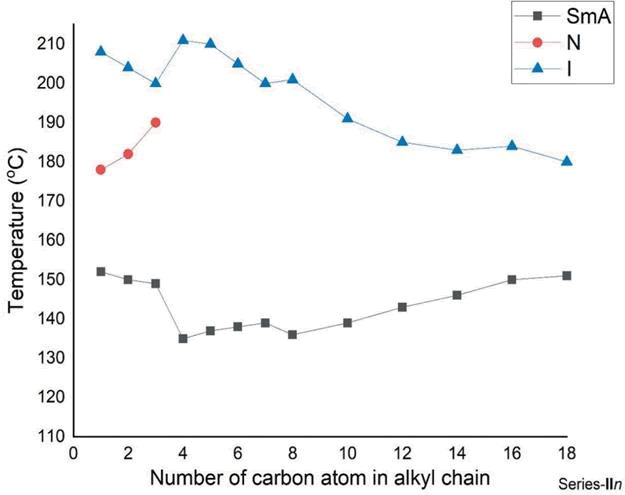
Figure 1: The phase behavior for series I.
| Compound | Transition | Peak temp./°C | ΔH/Jg-1 | ΔS/Jmol-1 K-1 |
| I10 | Cr-SmA | 100.84 | 47.16 | 0.1261 |
| SmA-I | 239.85 | 0.169 | 0.00032 |
Table 3: Enthalpy change, entropy change and normalized entropy of the series I10 by DSC.
Spectral data:
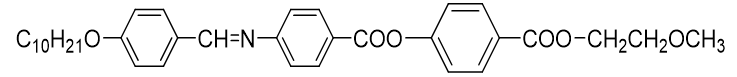
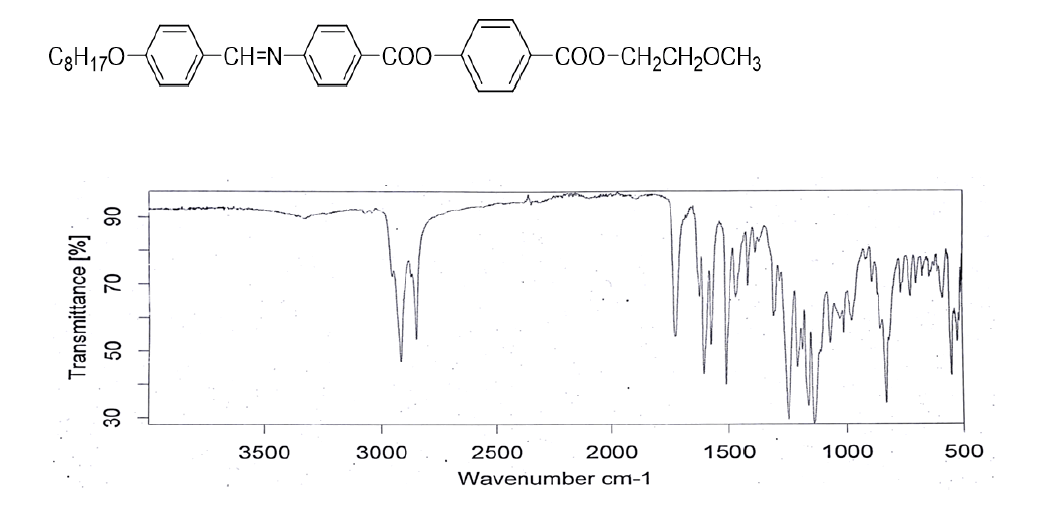
Figure 2: IR spectrum (KBr) Vmax/Cm−1: 2916, 2848, 1724 (-COO-), 1624 (-CH=N-), 1601, 1509, 1421, 1247, 1067, 851.
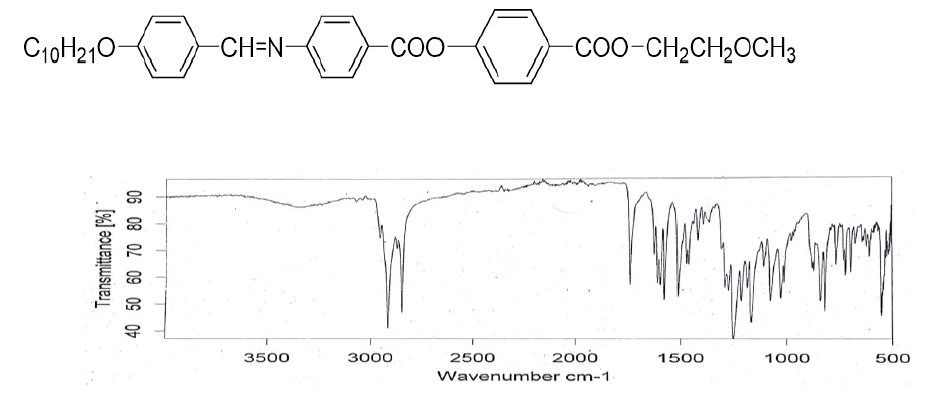
Figure 3: IR spectrum (KBr) Vmax/Cm−1: 2916, 2848, 1735 (-COO-), 1622 (-CH=N-), 1606, 1509, 1419, 1251, 1075, 838.
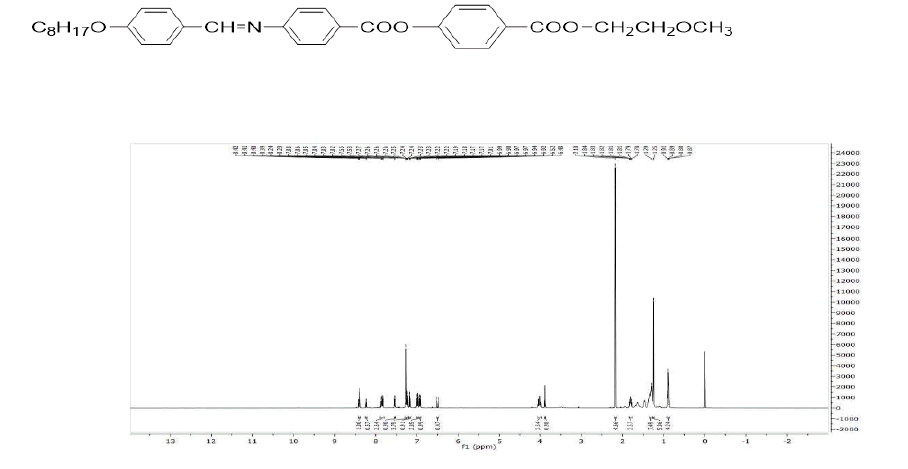
Figure 4: 1H-NMR spectrum (400 MHz, CDCl3) δ in ppm: 8.42 (s, 1H, -CH =N-), 8.20–8.16 (m, 4H, 2 × ArH), 7.86 (d, J=8.5 Hz, 2H, ArH), 7.58 (d, J=8.5 Hz, 2H, ArH), 7.28 (d, J=8.6 Hz, 2H, ArH), 6.98 (d, J=8.6 Hz, 2H, ArH), 4.43 (t, J=6.8 Hz, 2H, -COO-CH2-), 4.04 (t, J=6.4 Hz, 2H, Ar-O-CH2-), 3.77 (t, J=6.6 Hz, 2H, -CH2-O-), 1.74 (m, 2H, -CH2-), 1.44–1.26 (m, 6H, 7 X -CH2-), 0.93 (t, J=7.2 Hz, 3H, -CH3).
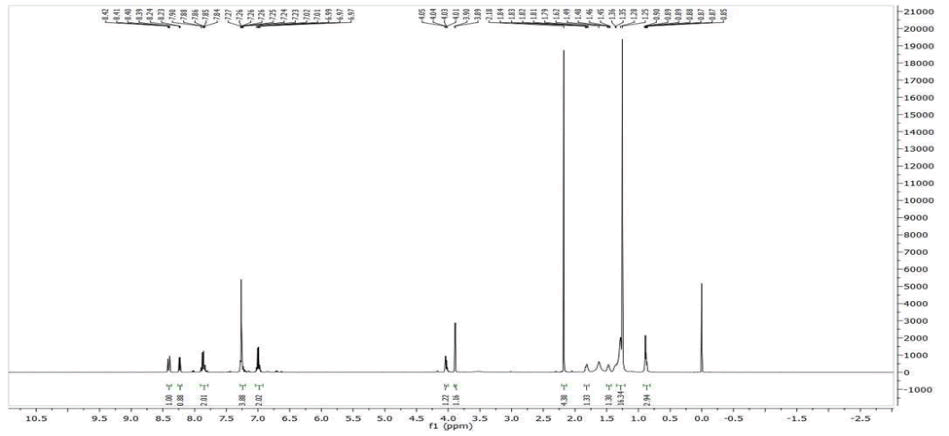
Figure 5: 1H-NMR spectrum (400 MHz, CDCl3) δ in ppm: 8.43 (s, 1H, -CH=N-), 8.20–8.16 (m, 4H, 2 × ArH), 7.86 (d, J=8.5 Hz, 2H, ArH), 7.58 (d, J=8.5 Hz, 2H, ArH), 7.28 (d, J=8.6 Hz, 2H, ArH), 6.98 (d, J=8.6 Hz, 2H, ArH), 4.43 (t, J=6.8 Hz, 2H, -COO-CH2-), 4.04 (t, J=6.4 Hz, 2H, Ar-O-CH2-), 3.77 (t, J=6.6 Hz, 2H, -CH2-O-), 1.74 (m, 2H, -CH2-), 1.44–1.26 (m, 10H, 7 X -CH2-), 0.93 (t, J=7.2 Hz, 3H, -CH3).
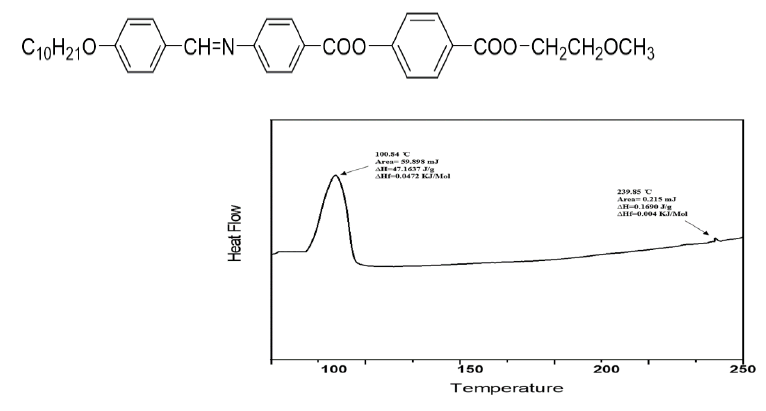
Figure 6: DSC Thermogram of series I (n=10).
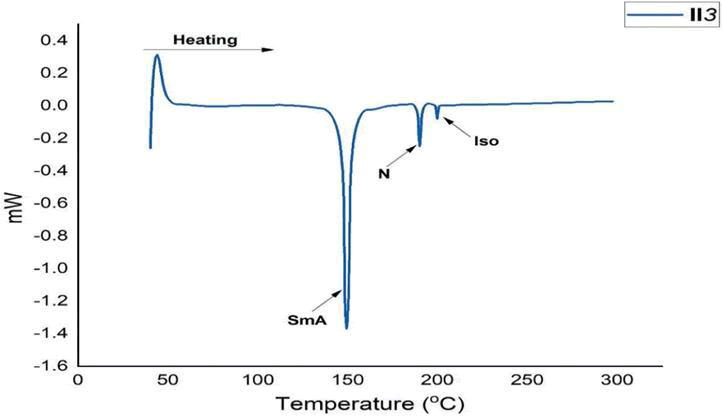
Figure 7: DSC Thermogram of series I (n=3).

Figure 8: Microphotograph of focal conic texture SmA and nematicmesophase (Series I; n=4) at 200ºC on cooling.

Figure 9: Microphotograph of focal conic texture nematicmesophase (Series I; n=2) at 221ºC on cooling.
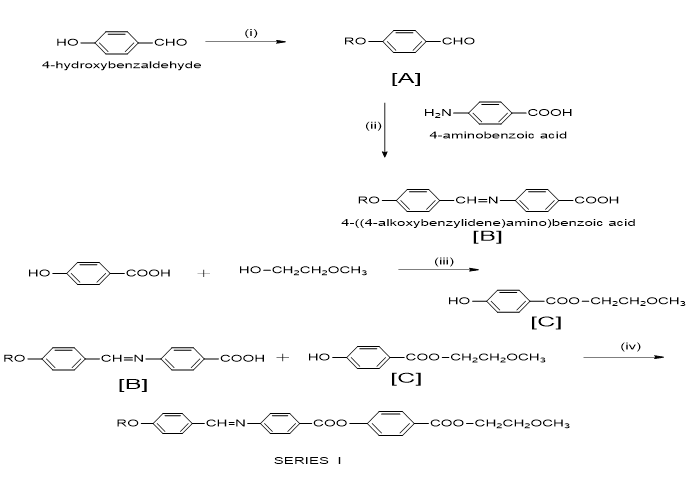
Figure 10: Synthetic route.
Note: Where, R=-CnH2n+1, n=1 to 8, 10, 12, 14 and 16.
Reagents and conditions: (i) R-Br, Dry K2CO3, dry acetone, (ii) P-Aminobenzoic acid, dry ethanol, acetic acid (iii)2-methoxy ethanol, Conc. H2SO4, (iv)DCC, DMAP, Dry THF.
| Compound | Cr-SmA | SmA-Iso | Mesophase range/°C | Commencement of smectic A phase |
| I | 134 | 221 | 87 | C1 |
| A | 142 | 211 | 69 | C1 |
| B | 66 | 104 | 38 | C1 |
| C | 93 | 158 | 65 | C2 |
| D | 158 | 238 | 80 | C1 |
Table 4: Comparison of the phase transition temperatures, smeticA mesophase range and comparative molecular structures of series I, A, B, C and D.
Conclusion
A new series of mesogenic homologues of Schiff base diesters with methoxyethyl tails have been synthesized. Series Iis exhibit smectic and nematic phases with triphenyl rings due to additional benzene rings and ester linkages. Strong methoxyterminated chains exhibit nematic and smectic mesophases. Studies have shown that the more polar methoxy end chain has a greater effect on the thermal stability of the mesophase than the chain end.
References
- Gray GW. Mol Cryst Liq Cryst. 1969;7(1):127-151.
- Gray GW, et al. Mol Cryst Liq Cryst. 1971;13(1):37-60.
- Dave JS, et al. Mol Cryst Liq Cryst. 1974;28(3-4):269-273.
- Lohar JM, et al. Mol Cryst Liq Cryst. 1981;70(1):279-287.
- Urs MS, et al. Mol Cryst Liq Cryst. 1982;72(7-8):227-231.
- Agrawal YK, et al. J Indian Chem Soc. 1990;67(5):369–370.
- Agrawal YK, et al. J Indian Chem Soc. 1997;74(10):689–691.
- Sadashiva BK. Mol Cryst Liq Cryst. 1976;35(3-4):205-213.
- Goodby JW. Liq Cryst. 2019;46(13-14):1901-1924.
[Crossref]
- Goodby JW, et al. Liq Cryst. 2015;42(5-6):593-622.
- Jankowiak A, et al. Liq Cryst. 2013;40(5):605-615.
- Demus D. Liq Cryst. 1989;5(1):75-110.
- Kohout M, et al. Liq Cryst. 2016;43(10):1472–1485.
- Jia YG, et al. Liq Cryst. 2017;44(3):526–537.
[Crossref]
- Luo CC, et al. Liq Cryst. 2017;44(14–15):2366–2378.
[Crossref]
- Zeng W, et al. Liq Cryst. 2019;46(12):1889–1898.
[Crossref]
- Imrie CT, et al. Chem Soc Rev. 2007;36:2096–2124.
- Imrie CT, et al. Liq Cryst. 2009;36:755–777.
- Vora RA, et al. Proc Indian Acad Sci Chem Sci. 2001;113(2):95–102.
- Prajapati AK, et al. Liq Cryst. 2004;31(6):889–894.
