Original Articles: 2021 Vol: 13 Issue: 1
Effect of Capparis Cartilaginea Leaves Extracts on Indomethacin-Induced Gastric Ulcer in Experimental Animals
Abdullah Ahmed Areqi1, Mohammed Gameel Al-Haddad2*, Bassam Abduh Ali2, Omar Abdullah Al-Tayeb1,Abdulsalam M Halboup3
1 Department of Pharmacology, Faculty of Pharmacy, University of Science and Technology, Hodaidah Branch, Yemen
2 Department of Pharmaceutics, Faculty of Pharmacy, University of Science and Technology, Hodaidah Branch, Yemen
3 Department of Clinical Pharmacy and pharmacy practice, Faculty of Pharmacy, University of Science and Technology, Hodaidah Branch, Yemen
Abstract
Background: Most parts of Capparis cartilaginea plant especially the leaves are used to treatment of many diseases such as skin inflammation, bruises, swellings, rheumatism, cough, indigestion, colics...etc. The leaves of C. cartilaginea are used traditionally in many areas of Yemen for heartburn/peptic ulcers, itching, shortness of breath and tumors.
Objective: To evaluate the potential anti-ulcer effect of C. cartilaginea leaves in experimental animals. Method: 15 male rabbits were divided into 5 groups. Each group contains 3 rabbits, Ulceration was induced in the groups II–V rabbits with indomethacin (40 mg/kg body weight, oral intubation) dissolved in distilled water, while group I was kept as a control and it was only given the vehicle oral dose of gum acacia in distilled water (2% w/w).
Results: The results showed that the leaves of C. cartilaginea have a significant effect (P<0.05) as anti-ulcer when compared to the indomethacin-induced gastric ulcer group (Group II). It was found that seven days post-ulcerative treatment with the C. cartilaginea leaves extracts was optimal for the effective healing of gastric ulcer.
Conclusion: Through this study, we conclude that the extracts of C.cartilaginea leave have anti-ulcerogenic activity and they can be used to treat gastric ulcer.
Keywords
Capparis cartilaginea, Ulcerogenic; Indomethacin; Gastric Ulcer; Omeprazole; Rabbits
Introduction
Many of the pharmaceutical drugs in use are derived from plants directly or indirectly, it is obvious that at least some plants contain compounds with pharmacological activity that can be harnessed as medicinal agents; while few would disagree with that proposition, there are many who persist in referring to herbal medicines (along with other “alternative remedies”) as unproven and therefore of little or no clinical value; increasingly, the public particularly the medical establishment are demanding herbalists and other complementary therapists provide scientific evidence for the efficacy and safety of their practices; while this is an admirable objective, it cannot be achieved overnight, given the complexities of the herbs themselves, the variety of formulas and prescribing methods available and the difficulties in adapting medical models to the herbal practice [1].
Capparis is a medicinal plant coined by Theophrastus (4th century BC) and endorsed by Dioscorides (1st century AD); it seems to have come into wide use after the spread of the Arab culture in the Middle Ages; the genus Capparis was created by Linnaeus with the description of Capparis spinosa L. and other Capparis species [2]. Capparis comprises around 250 species distributed in tropical and subtropical zones of southern America, Europe, Africa, Madagascar, Asia, Australia, and the Pacific Islands. The family name of caper plants is Capparidaceae or Capparaceae [3].
Caper plants are small shrubs, and may reach about one meter upright; however, uncultivated caper plants are more often seen hanging, draped and sprawling as they scramble over soil and rocks [4].
Chemistry of caper plants: the flower buds contain a glycoside, rutin, which on acid hydrolysis yields rhamnose, dextrose and quercetin; flower buds also contain about 4% pentosans on a dry weight basis, rutic acid, pectic acid and saponin; caper seeds yield about 35% pale yellow oil containing palmitic, stearic, oleic and linoleic acids; the root bark contains rutic acid and a volatile substance with a garlic odour; a series of isomers of the compound cappaprenols have been isolated from Capparis. Glucobrassicin, neoglucobrassicin and 4-methoxyglucobrassiin were identified in the roots by High-Performance Liquid Chromatography (HPLC) [5].
Capparis cartilaginea Decne. (Synonyms include C. galeata Fres, C. inermis Forsskal, C. sinaica Veillard, C. spinosa var. galeata (Fres.) Hook. F. and Thorns, C. uncinata Edgew) [5] has many common names. These common names are in Arabic: felfel-jibbel, goah, goah-kulul, lassaf, kabar (Egypt); lassaf, lusef (Plant), 'aslub, albelib (fruit), làtssaf, nutssàf (Saudi Arabia); lúsfeh (Plant), àlbelib, àslub (fruit) (Dhofar); lattssaf, laşaf, nişaf (Yemen); in Hebrew: tsalaf şhusi; in Jibbali: lósed, aselib (fruit) (Dhofar); in India: karat; in Iran (Balouchistan): gorilimbuk blatter; in Kenya: chepkogh, chepteretwa, gorra, ilngorochi, leaches, lokapilak, mtunguru, mbaruti, olatunde, qadhu; and in Somalia: goah, goah-kulul, goh, gombor [6].
Experimental Section
Water samples were collected from about 15 cm below the surface of water in wide mouthed screw capped airtight and opaque polythene containers. The samples were collected fortnightly from June 2014 to May 2016. The samples were collected on 1st and 15th of every month around 9:00 a.m. The atmospheric and water temperature was recorded with the help of digital portable kit. The digital portable kit is an excellent instrument for field operations. The date of humidity and rainfall was collected from meteorological department Chikalthana, Aurangabad. The pH of the sample was recorded with “Systronic portable pH meter”.
Dissolved oxygen, Acidity, total alkalinity, hardness, Nitrogen, Sulphate, phosphates, total solids etc. has been determined as per the procedures given in literature. There are three seasons during a year in this region, namely summer (February to May), Monsoon (June to September) and winter (October to January). The summer season is characterized by clear sky with relatively longer duration of day and more intensity of light. During monsoon, the sky is cloudy humidity is relatively more and temperature is moderate to high. The winter has relatively brighter days with clean sky and lesser during of day. As all ecological factors are influenced by the duration and intensity of sunlight, humidity, atmospheric and water temperature, various physico chemical variables were studied season wise and at the selected sample station, during the present study over a period of two years.
Description of C.cartilaginea Decne. as shown in Figure 1 is perennial shrub with straggling or scrambling stems, spiny; leaves 2-6 it 2-6 cm, ovate to orbicular, somewhat fleshy, apex emarginated with a spine below the apex; flowers white, up to 4 cm; many stamens, exserted; fruit 3-5 × 2-3 cm, ellipsoid to oblong, red, many-seeded; seeds embedded in pulp[7]
The local medicinal uses of C.cartilaginea decne.: the fruits used for rheumatism; veterinary (fevers); the leaves and stems/shoots used for skin inflammation; bruises; swellings; rheumatism, joint inflammation; knee problems; tendinitis; sprains; muscular contractions; paralysis of body members; headache; earache; for eye disease (humans, cattle); cough; indigestion, colics; childbirth, after childbirth for pains and as an antiseptic; snakebites (pain, inflammation); veterinary medicine (parasites and ticks in livestock; cattle fevers; common cold in newborn goats; for sickly camels and goats; first causes severe diarrhea, then improvement of appetite, good condition, and milk increased in quantity and quality); and the roots used for dermatitis; skin ulcers; wounds [6].
The leaves of C. cartilaginea decne Are shown in Figure 2 and used traditionally for heartburn/peptic ulcers [8], itching, shortness of breath, head cold and tumors [9,10].
For treatment of leaves and young stems, they are crushed in water and heated; the solution is strained and applied on bruises, snakebites, swellings, on the forehead for headaches and on skin rash to reduce itching, leaves are crushed in water and the water boiled, strained and used to wash the new mother after childbirth; the juice is considered to be antiseptic; the juice extracted from the leaves is applied on swellings and with vinegar added is used as eardrops to treat deafness; a paste made of leaves and stems is applied on swollen and painful joints, muscle pain and paralyzed limbs [9].The leaf of C. cartilaginea decne consists of many phytochemicals such as carbohydrates, saponins, polyphenols, flavonoids, tannins, triterpenes, sterols, amino acids and proteins; the flavonoid and saponin content are 5.1% and 1.8% respectively [11,12].
Peptic ulcer disease usually occurs in the stomach and proximal duodenum, and the predominant causes in the United States are infection with Helicobacter pylori and use of nonsteroidal anti-inflammatory drugs; and the symptoms of peptic ulcer disease include epigastric discomfort (specifically, pain relieved by food intake or antacids and pain that causes awakening at night or that occurs between meals), loss of appetite, and weight loss [13].
Prostaglandins (PGs) are known to protect the gastric mucosa against injury caused by a variety of necrotizing agents; furthermore, PGE2 stimulates the secretion of gastric mucus, bicarbonate, and surfactant-like phospholipid; nonsteroidal anti-inflammatory drugs (NSAIDs) such as indomethacin, acetylsalicylic acid (ASA) may cause gastric mucosal injury in humans and experimental animals by inhibiting the biosynthesis of PGs, thus acting as cyclooxygenase (COX) inhibitors; these data suggest that PGs play a physiological role in maintaining the integrity of gastric mucosa; however, only limited information is available regarding the site of synthesis and regulation of endogenous PGE2 in the stomach; the regulation of PG production during the acute stage and the subsequent repair process of gastric lesions are also not understood at present[14].
The integrity of the gastric mucosa is dependent on a balance between luminal aggressive factors (such as acid and pepsin) and the maintenance of the protective mucosal barrier; the damage to the gastric mucosa results when this balance is disrupted by either an increase in aggressive factors or disruption of the protective gastric mucosal barrier; the chronic therapy with non-steroidal anti-inflammatory drugs (NSAIDs) is thought to damage the gastric mucosa by two mechanisms:
(i) By direct local injury and (ii) By systemic inhibition of cyclooxygenase resulting in a relative deficiency of prostaglandins [2].
Gastric mucus is an important protective factor for the gastric mucosa and consists of a viscous, elastic, adherent and transparent gel formed by 95% water and 5% glycoproteins that covers the entire gastrointestinal mucosa; Moreover, mucus is capable of acting as an antioxidant, and thus can reduce mucosal damage mediated by oxygen free radicals; the protective properties of the mucus barrier depend not only on the gel structure but also on the amount or thickness of the layer covering the mucosal surface, so a decrease in gastric mucus renders the mucosa susceptible to injuries induced by gastric acid [13].
The acute lesions of the gastric mucous membrane is associated with the rupture of the hydrophobic surface of the membrane; injury and exfoliation of the epithelium surface with loss of barrier and electrical function: deep injury of the mucosa layer, including vascular endothelial cells and cell proliferation zone; when there is damage to the vascular endothelium, a decrease of blood flow may occur, which reduces the oxygen supply and consequently the nutrients transport; all these events result in the appearance of erosion and ulceration in the mucous membrane [6].
Omeprazole is proton pump inhibitor drug used to treatment of peptic ulcer. It inhibits the final step in the formation of hydrochloric acid by blocking the enzyme H+, K+-ATPase; it is a highly effective inhibitor of acid secretion and promotes rapid healing of duodenal ulcer and reflux esophagitis.
C.cartilaginea leaf is used as a traditional medicine in many governorates of Yemen. It is used for treatment of peptic ulcers [11] and treatment of tumors [9,10]. However, there are not enough documented or published papers on such effects available.
Due to the lack of pharmacological information and clinical application of C. cartilaginea on the treatment of gastric ulcer, the main information available present at the following:
It was investigated the benefits of traditional uses of this plant as anticancer, antimicrobial and antioxidant in vitro. Other study investigated the methanolic extract of same plant but with another species (capparis zeylanica) has antiulcer activity on experimental animals [12]. As well as there is a study identify the effect of polyphenolic extracts of the same plant but with another species (Capparis Spinosa L.) where found to reduce serum glucose in alloxan-induced diabetes mellitus in female rats. Furthermore another study find the antioxidant effect of this plant by DPPH compared to standard quercetin of similar concentration.
The aim of this study is to evaluate the potential anti-ulcer effect of C. cartilaginea leaves in experimental animals through measuring different parameters like: ulcer index, cortisol level, Aspartate Aminotransferase (AST) level also called Glutamic Oxaloacetic Transaminase (GOT), alkaline phosphatase (ALP) level and chloride level.
Materials and Methods
Indomethacin was purchased from Sigma (Egypt), Omeprazole from Cipla (India), Ketamine from Rotexmedica (Germany). Leaves of C. cartilaginea were collected from Almesbah - Wosab - Dhamar – Yemen.
Capparis cartilaginea leaves were obtained freshly from Almesbah - Wosab - Dhamar - Yemen. The leaves were thoroughly washed several times using normal tap water; then this was continued by distilled water to remove the impurities. 20 g of fresh leaves were boiled in 100 ml filtered water. After 10 min, leaves were crashed and boiled again for 5 minutes. The prepared solution was initially filtered through normal filter paper mesh so that the leafy materials could be filtered out; then the extract was filtered through Whatman filter paper No. 1. The filtered extract was stored in the refrigerator at 4ºC.
Extract of Dried Leaves of C. Cartilaginea
Dried leaves of C. cartilaginea were extracted traditionally as the people do when they want to use these leaves for therapy, they were crushed, then one teaspoonful (tsp) of the crushed leaves was taken and placed in 50 ml of water; the mixture was stirred for 5 minutes.
Experimental Animals
15 male rabbits, aged (12-15 ± 2 months) and weighing (1500 g ± 100 g) were obtained from animal house. They were acclimatized to the laboratory conditions 2 days before the start of the treatment. The animals were starved 24 hours, and only water was given to them prior to the experiment. The animals were weighted and given specific numbers and marks.
Ulcer Induction
Indomethacin was used to induce ulcer in the animals, it act as an NSAID (Non-steroidal anti-inflammatory drug). Gastric ulceration was induced in the animals by giving them a single oral dose of indomethacin 40 mg/kg body weight in 10 ml of distilled water; they were deprived of food but had free approach to water 24 hours prior to ulcer induction. Before starting the treatment a rabbit were sacrificed after 4 hours of indomethacin induction as a pilot study to make sure that Indomethacin has induced gastric ulcer in this rabbit.
Study Design
The protocol we followed in the division of rabbits was obtained from [4] with modifications as the following:
Rabbits were divided into 5 groups. Each group contains 3 rabbits.
Group I: Rabbits serving as the normal control received only the vehicle oral dose of gum acacia in distilled water (2% w/w) 10 ml per rabbit. Ulceration was induced in the groups II–V rabbits with indomethacin (40 mg/kg body weight, oral intubation) dissolved in 10 ml of distilled water.
Group II: Rabbits were given the vehicle (10 ml per rabbits) only for 7 days.
Groups III: Rabbits were received omeprazole (20 mg/kg body weight) once daily by oral intubation starting from 4 hours after the indomethacin administration.
Groups IV: Rabbits were received the extract of fresh leaves of C. cartilaginea (10 ml of the extract, about 200 mg).
Groups V: Rabbits were received the extract of dried leaves of C. cartilaginea (10 ml of the extract).
The treatment was continued for 7 days, after that the rabbits were sacrificed and the tissue samples of the stomach were collected after the 7th day of the treatment to compare between them.
Collection of Blood Samples
At the end of experimental period the blood was collected and placed in a sample bottle to clot. After clotting the blood was centrifuge 3000 rpm and serum was separated in different bottles and kept in the freezer prior to use. The serum obtained was used for the assay of biochemical parameters. These biochemical parameters were cortisol, AST, ALP and chloride. After the blood collection the rabbits were kept unconscious using chloroform by inhalation and ketamine HCl (100 mg/kg, IP) in order to be sacrificed.
Ulcer Measurement
Fresh stomach from each rabbit was taken and opened along the greater curvature Figure 3. The ulcer index (UI) was calculated macroscopically according to this method:
Ulcer index=10/X
Where X=Total mucosal area / Total ulcerated area.
The number of ulcers were counted using a magnifying lens and then measured with a caliper to determine the diameter. The percentage of inhibition with omeprazole and C. cartilaginea groups was calculated according to the following formula:
Where UI standard for ulcer index
Data Analysis
Data were summarized as means ± SEM. Furthermore, paired sample T-test was used to conduct the significance of association using SPSS Program, version 20. Differences were considered significant at P-values of less than 0.05.
Results
Effect of the Drugs on Gastric Ulcer Healing In Rabbits
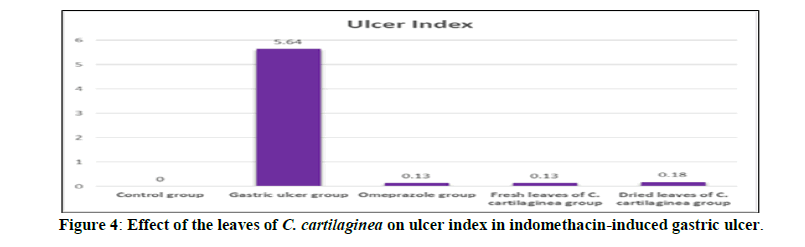
Based on the results obtained, the leaves of C. cartilaginea showed a significant effect (P<0.05) as anti-ulcer when compared to the indomethacin-induced gastric ulcer group (Group II). Table 1 and Figures 4, 5 shows the effect of the leaves of C. cartilaginea on ulcer index. It was found that seven days post-ulcerative treatment with the C. cartilaginea leaves extracts was optimal for the effective healing of gastric ulcer.
| Groups | Ulcer Index | inhibition% |
|---|---|---|
| Control Group | - | - |
| Gastric Ulcer Group | 0.04 ± 5.64 | - |
| Omeprazole Group | *0.003 ± 0.13 | 97.87 |
| Fresh Leaves of. C Cartilaginea Group | *0.003 ± 0.13 | 97.7 |
| Dried Leaves of. C Cartilaginea Group | *0.003 ± 0.18 | 96.81 |
Table 1: Effect of the leaves of C. cartilaginea on ulcer index and percentage of inhibition in indomethacin-induced gastric ulcer
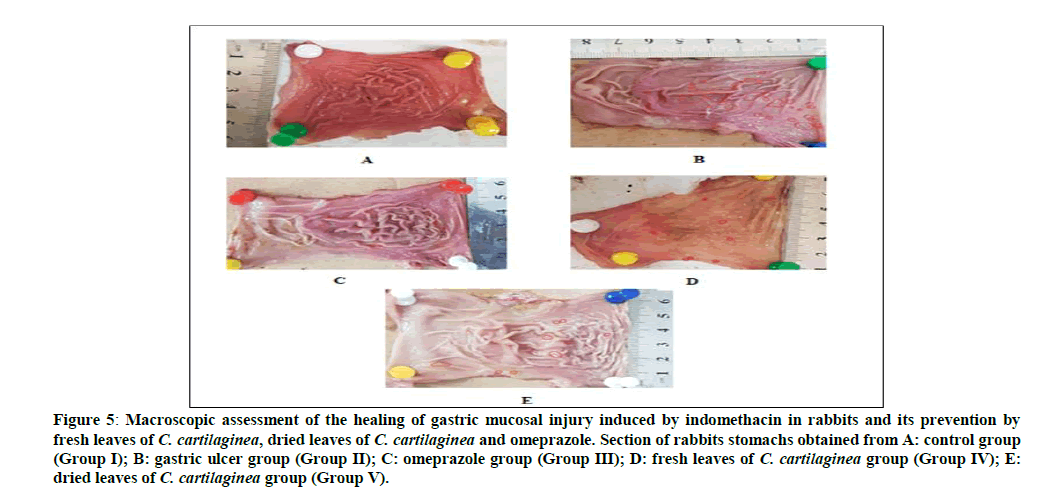
Figure 5: Macroscopic assessment of the healing of gastric mucosal injury induced by indomethacin in rabbits and its prevention by fresh leaves of C. cartilaginea, dried leaves of C. cartilaginea and omeprazole. Section of rabbits stomachs obtained from A: control group (Group I); B: gastric ulcer group (Group II); C: omeprazole group (Group III); D: fresh leaves of C. cartilaginea group (Group IV); E: dried leaves of C. cartilaginea group (Group V).
Effect of the Drugs on the Parameters in Indomethacin-Induced Gastric Ulcer in Rabbits
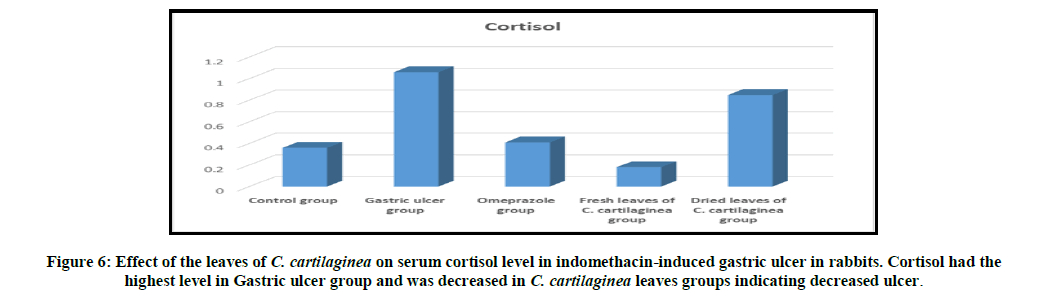
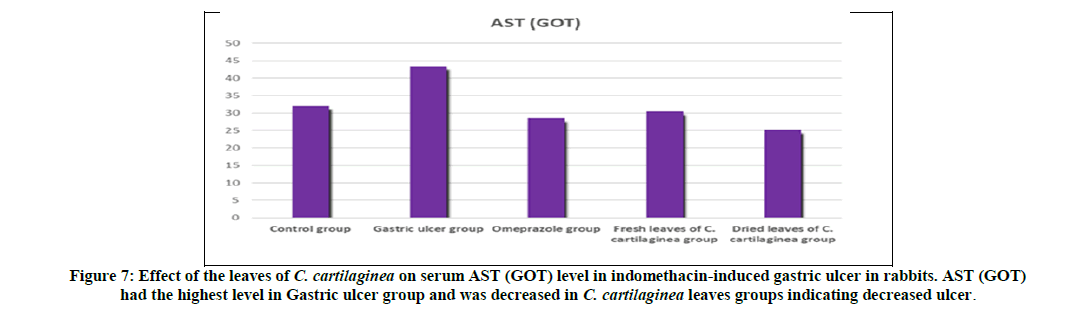
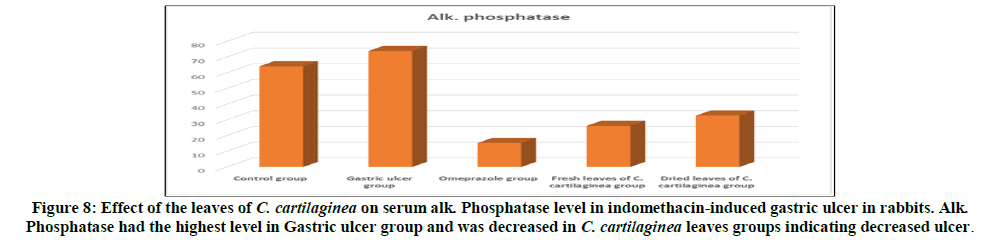
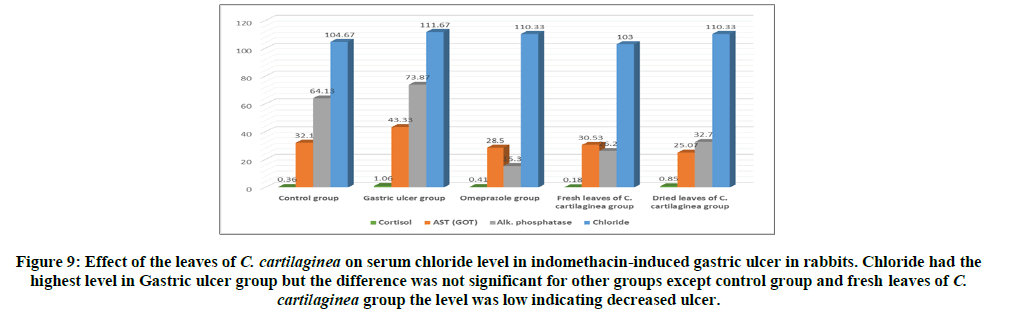
Rabbits treated with indomethacin (Group II) significantly had increased serum cortisol, AST (GOT), alk. phosphatase and chloride levels when compared to the normal control group (Group I). The leaves of C. cartilaginea showed a significant decrease (P<0.05) in the levels of serum cortisol, AST (GOT), alk. phosphatase and chloride when compared to the ulcerated control rabbits (Group II) as shown in Table 2 and Figures 6, 7, 8, 9 and 10.
| Parameter/Group | Cortisol (µg/dl) | AST (GOT) (U/L) | Alk. Phosphatase (U/L) | Chloride (mmol/L) |
|---|---|---|---|---|
| Control Group | 0.36 ± 0.02 * | 32.10 ± 0.75 * | 64.13 ± 1.91 | 104.67 ± 0.88 |
| Gastric Ulcer Group | 1.06 ± 0.05 # | 43.33 ± 1.10 # | 73.87 ± 1.41 | 111.67 ± 1.45 |
| Omeprazole Group | 0.41 ± 0.01 * | 28.50 ± 1.27 * | 15.30 ± 1.54 *# | 110.33 ± 0.88 |
| Fresh Leaves of C. cartilaginea Group | 0.18 ± 0.02 *# | 30.53 ± 1.44 * | 26.20 ± 1.54 *# | 103.00 ± 0.58 * |
| Dried Leaves of C. cartilaginea Group | 0.85 ± 0.04 # | 25.07 ± 0.96 *# | 32.70 ± 1.04 *# | 110.33 ± 0.88 |
Table 2: Effect of the leaves of C. cartilaginea on serum cortisol, AST (GOT), alk. Phosphatase and chloride levels in indomethacininduced gastric ulcer in rabbits
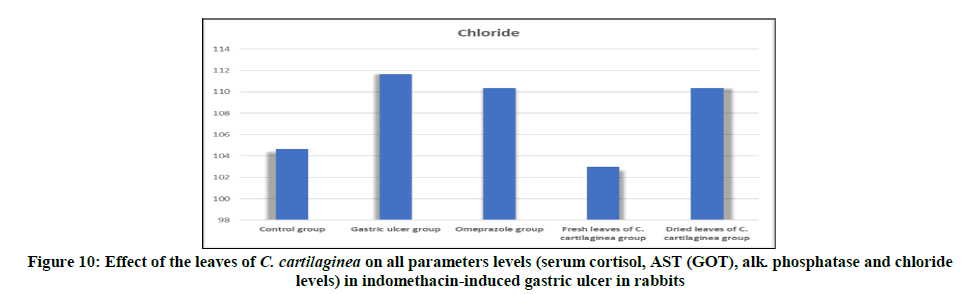
Figure 9: Effect of the leaves of C. cartilaginea on serum chloride level in indomethacin-induced gastric ulcer in rabbits. Chloride had the highest level in Gastric ulcer group but the difference was not significant for other groups except control group and fresh leaves of C. cartilaginea group the level was low indicating decreased ulcer.
Discussion
Treatment of rabbits with indomethacin produced lesions in the gastric mucosa (Group II) resulting in substantial ulcer index as shown in Table 1 and Figures 4,5. This was reduced by 97.87% for the omeprazole group (Group III) compared to the ulcerated group (Group II) due to healing, also treatment with the extracts of fresh leaves and dried leaves of C. cartilaginea reduced the ulcer index by 97.70% and 96.81% respectively, that means that leaves of C. cartilaginea have efficacy in treatment of gastric ulcer. The effects of indomethacin intake alone and following administration of the drugs on the serum levels of cortisol, AST (GOT), alk. Phosphatase (ALP) and chloride in rabbits are shown in Table 2 and Figures 6, 7, 8, 9 and 10.
In the current study, significant rise in serum adrenocorticotropic hormone (ACTH) level in gastric ulcer has been recorded. Gastric juice cortisol concentration increases after stimulation of ACTH [15]. In this study indomethacin administration markedly stimulated cortisol in the indomethacin induced ulcer group (Group II) and the cortisol was elevated when compared to the ulcerated control rabbits (Group I). While in the treated groups (Group III, IV and V) the cortisol was decreased after seven days of treatment and that may be due to healing of the gastric ulcer.
There is a study has found that the enzymatic activity of aspartate aminotransferase (AST) and alkaline phosphatase (ALP) increases in peptic ulcer disease [5]. Also another study has found that the enzymatic activity of alkaline phosphatase (ALT) is significantly high throughout the ulcerous zone in case of gastric ulcer [11]. The levels of serum AST and ALP in both treated groups with C. cartilaginea leaves (Group IV and V) and negative control group (Group I) were significantly decreased when compared with the indomethacin induced ulcer group (Group II) suggesting a healing effect of the gastric ulcer by the C. cartilaginea leaves (Group IV and V) and also omeprazole (Group III).
The concentration of chloride tends to rise and fall with the fluctuations in the rate of secretion of gastric juice [16]. The increased level of chloride in the indomethacin induced ulcer group (Group II), omeprazole group (Group III) and dried leaves of C. cartilaginea group (Group V) may be as a result of gastric juice secretions. Also may be as a result of higher buffering capacity of bicarbonate. However in the control group (Group I) and fresh leaves of C. cartilaginea group (Group IV) chloride level tend to be normal and that means that the fresh leaves of C. cartilaginea (group IV) may have various mechanisms in the treatment of gastric ulcers.
Conclusion
The leaves of C. cartilaginea produces anti-ulcer activity similar or close to a standard drug omeprazole, and the mean ulcer indexes of the drugs were found to be statistically significant (P<0.05). Also C. cartilaginea leaves showed a statistical significant decrease in cortisol, AST and ALP activity (P<0.05) when compared to indomethacin-induced gastric ulcer rabbits (Group II). Therefore, we conclude that the extracts of C. cartilaginea leave have anti-ulcerogenic activity and they can be used to treat gastric ulcer.
Recommendation
We recommend to conduct further clinical studies with more than one species and large numbers of experimental animals. Also we recommend taking into account the same conditions of extraction for both fresh leaves and dried leaves of C. cartilaginea with accurate doses for them in order to approve and expand these findings.
Acknowledgement
The authors want to thank the 5th year pharmacy students at university of science and technology 2018 batch, for funding a large part of this research.
References
- KV Peter. Handbook of herbs and spices, 2nd Edition, Cambridge England: Woodhead Publishing Limited, Boca Raton, Boston, New York and Washington. 2006, 22-27.
- S Manikandaselvi; V Vadivel; P Brindha. Asian J Pharm Clin. Res. 2016, 9, 123-126.
- K Ramakrishnan; RC Salinas. Peptic ulcer disease, American family physician. 2009, 76(7), 1006.
- F Benakashani; AR Allafchian; SA Jalali. J Modern Science. 2016, 2(4), 251-258.
- U Quattrocchi. CRC world dictionary of medicinal and poisonous plants: common names, scientific names, eponyms, synonyms, and etymology, 1st Edition, CRC press London and New York. 2016, 786.
- Quattrocchi. CRC world dictionary of medicinal and poisonous plants: common names, scientific names, eponyms, synonyms, and etymology, 1st Edition, CRC press London and New York. 2016, 786.
- M Lee; SM Kallal; M Feldman. Alimentary pharmacology & therapeutics. 1996, 10(4), 571-576.
- C Inocencio; D Rivera; MC Obón; F Alcaraz; JA Barreña. Annals of the Missouri Botanical Garden. 2006, 93(1), 122-149.
- HK Kley; H Peerenboom; G Strohmeyer; HL Krüskemper. Digestive diseases and sciences. 1983, 28(6), 494-501.
- NA Galib; SK Algfri. J Medicinal Plants. 2016, 4(5), 280-286.
- MG Repetto; SF Llesuy. Brazilian J medical and biological research. 2002 35(5), 523-534.
- SA Ghazanfar. Handbook of Arabian medicinal plants. V1 Boca Raton, Ann Arbor, London, Tokyo. 1994, 73-74.
- A Pengelly. Constituents of medicinal plants: an introduction to the chemistry and therapeutics of herbal medicine, 2nd Edition, CABI Publishers, Australia: Allen & Unwin. 2004, 1.
- RA Mothana; U Lindequist; R Gruenert; PJ Bednarski. BMC Complementary and Alternative Medicine. 2009, 9(1), 7.
- M Basbag; O Toncer; S Basbag. J environmental biology. 2009, 30(4), 621.
- S Bhattacharya; SR Chaudhuri; S Chattopadhyay; SK Bandyopadhyay. J Clinical Biochemistry and Nutrition. 2007, 41(2), 106-114.
- M Boghori; M Aghamaali; R Sariri; F Mohamadpour; H Ghafouri. J oral biology and craniofacial research. 2014, 4(1), 24-29.
- CAD Carvalho; KM Fernandes; SLP Matta; MBD Silva; LLD Oliveira; CC Fonseca. Arquivos de gastroenterologia. 2011, 48(4), 276-282.
- DA Elebeedy; E Maksoud; ZM Yasser; YM Salah. The Egyptian J Medical Microbiology. 2017, 26(3), 37-43
- GG Ivanov. Voprosy meditsinskoi khimii. 1978, 24(2), 206-210
- R Karanayil; N Barij; A Rajasekaran. Avicenna J Med Biotech. 2011, 3(1), 31-35.
- D Khan; SS Shaukat; SV Ali. J Biol Res. 2015, 3(1), 19-29
- W Kipkore; B Wanjohi; H Rono; G Kigen. J ethnobiology and ethnomedicine. 2014, 10(1), 24.
- EP Lansky; M Paavilainen; S Lansky. Caper: the genus Capparis. V. 2013, 39.
- HI Mizuno; CH Sakamoto; KO Matsuda; K Wada, TO Uchida; HI Noguchi.Gastroenterology. 1997, 112(2), 387-397.
- NA Galib; SK Algfri. J Medicinal Plants Studies. 2016, 4(5), 280-286.
- K Ramakrishnan; RC Salinas. Peptic ulcer disease, American family physician. 2009, 76(7), 1006.
- SK Oudah; MH Raid; Al-Salih; SH Gusar. International Journal of Scientific & Engineering Research. 2014, 5(5), 1561-1575.
- SE Kotob; AH Sayed; SH Mohamed; HH Ahmed. Asian Journal of Pharmaceutical and Clinical Research. 2014, 11(1), 381-389.
- JA Strong; D Cameron; MJ Riddell. Experimental physiology. 1960, 45(1), 1-11.
- WRS Uma; MN Shehu. J Pharmacognosy and Phytochemistry. 2016, 5(1), 80-84.
- OV Virchenko; TM Falalyeyeva; TV Beregova; SV Maryana. Research J Pharmaceutical Biological and Chemical Sciences. 2015, 6(1), 249-259.