Original Articles: 2021 Vol: 13 Issue: 1
Assessment of Pseudomonas aeruginosa Resistance to Antibiotic among Infected Patient in Hodeidah City
Mohammed Gameel Al-haddad1, Zaid Ali AL-Gadary2*, Aisha Nashwan Al-Romaih2, Safa Adel Al-Athwari2
1Department of Pharmaceutics, Faculty of Pharmacy, University of Science and Technology, Hodeida Branch, Yemen
2Department of Laboratory, Faculty of Medicine and Health Sciences, University of Science and Technology, Hodeida Branch, Yemen
Abstract
Background: All pseudomonads are widespread in nature. The most important species in this group from a medical point of view is Ps. aeruginosa, which causes infections in person with immune defects. Objective: Determine the antibiotic that are effective to Ps.aeruginosa among infected Patient in Hodeidah City , know Ps aeruginosa resistance to antibiotic among infected patient in Hodeidah City and Provide a medical information about these bacteria and resistant to antibiotic.
Methods: The study was performed at microbiology department of a local Al-Hodeidah hospitals and laboratory Hodeidah city, Yemen. All the samples were collected during the study period, from May 2020 and ending in Augusta's 2020. All specimens were transported within one hours of collection to UST microbiology laboratory for processing, culture and identification. Ten isolates of Ps. aeruginosa were isolated from different clinical specimens and fully characterized by standard bacteriological procedures. Antimicrobial susceptibility pattern of each isolates was carried out by the Kirby-Bauer disk diffusion method.
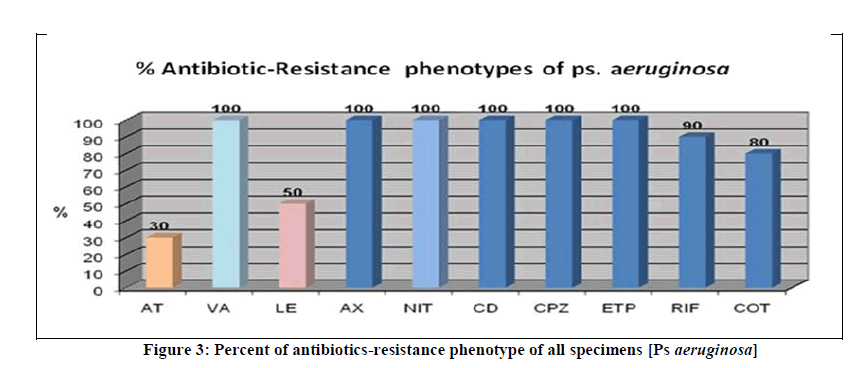
Results: The isolate pathogen showed highly resistant to six types of antibiotics which are Vancomycin, Ampicillin,cloxacilli, Nitrofurantion, Clindamycin, Cefoperazon, Ertapenam with a percentage of [100%], Rifampin [90%] and Trimethoprim/sulphamethoxazole [80%] while Aztreonam [70%] and Levofloxacin [50%] are considered to be sensitive.
Conclusions: The result confirmed the occurrence of drug resistance strains of Ps. aeruginosa. Vancomycin, Ampicillin/cloxacilli, Nitrofurantion, Clindamycin, Cefoperazon, Ertapenam and Rifampin were found to be the most effective antimicrobial drugs. It therefore calls for a very judicious, appropriate treatment regimens selection by the physicians to limit the further spread of antimicrobial resistance Ps. aeruginosa.
Keywords
Antimicrobial Susceptibility; Pseudomonas aeruginosa; Multi Drug-Resistant; Aztreonam
Introduction
Pseudomonads are Gram-negative, aerobic, rod-shaped bacteria with widespread occurrence in nature, Pseudomonas aeruginosa is clinically significant and opportunistic pathogen that causes infections in hospitalized patients. In addition, most Pseudomonas species have intrinsic resistance to many antibiotics and ongoing emergence of new resistance can be developed after commonly prescribed antimicrobial agents [1].
The pathogenesis of Pseudomonas aeruginosa infections is complex. The organism can use its attachment pili to adhere to host cells. The relevant virulence factors are: exotoxin A, exoenzyme S, cytotoxin, various metal proteases, and two types of phospholipase C [2]. The lipopolysaccharide of the outer membrane also plays an important role in the pathogenesis. Pseudomonas aeruginosa infections occur only in patients with weakened immune defense systems, notably pneumonias in cystic fibrosis, colonization of burn wounds; endocarditis in drug addicts, postoperative wound infection, urinary tract infection, sepsis, P. aeruginosa frequently contributes to nosocomial infections [3]. The resistant strains of Pseudomonas aeruginosa that cause nosocomial infections contributes substantially to the morbidity and mortality of hospitalized patients [4]. Despite the availability of a variety of effective antimicrobial agents, treatment of pseudomonal aeruginosa is often challenging antimicrobial resistance is a growing problem worldwide, especially in hospitals, where resistant organisms are often first detected in ICUs [5]. Multiple resistances to anti-bacterial agents present a therapeutic problem [6].
It was also reported frequently from patients undergoing chemotherapy for neoplastic diseases [7]. The variations of antibiotic protocols in clinics or in regions result in the different resistance profiles [8]. It is, therefore, the goal of this study to determine the antibiotic that are effective to Ps. aeruginosa among infected Patient in Hodeidah City and know Ps aeruginosa resistance to antibiotic among infected patient in Hodeidah City, Yemen also to evaluate its susceptibility against certain antibiotics, as limited work has been previously conducted on this subject.
Materials and Methods
Specimens Collection
In this study, bacteriology specimens was taken from patients with wound infection and urinary tract infection, were collected according to the standard method of Baily and Scott Diagnostic microbiology [9]. All specimens were transported within one hours of collection to UST microbiology laboratory for processing, culture and identification. During a period of three months, starting in May 2020 and ending in August 2020.
Study Design and Area
Cross sectional study was taken from Al-Hodeidah hospitals and laboratory Hodeidah city –Yemen
Bacteriological Examination Culture Media
Differential, enriched and/or selective media were used for culture the etiological agents [Pseudomonas aeruginosa]. The collected specimens were inoculated on blood ager, chocolates ager and MacConkey ager.
Isolation and Identification of Bacterial Isolates
Standard method of isolation and identification were used to isolate Pseudomonas aeruginosa bacteria in pour culture, than identified by microscopy Gram stain reaction, proper enzymes detection and biochemical reactions.
Antibiotics Susceptibility Test
The disk diffusion method was used for susceptibility of isolated bacteria to different antimicrobial agent. At least 4 to 5 well isolated colonies of the same morphological type from an ager plate were selected with a wire loop and transferred to tube containing 4 to 5 ml of sterile physiological saline or nutrient broth. The turbidity of this preparation was compared to the turbidity of barium chloride. A sterile swab was used to inoculate the broth on a plate of Muller Hilton ager. The swab was streaked evenly over the surface of the medium in three directions, rotating the plate approximately to ensure even distribution. The plate then left for 5 minutes for drying. Using sterile forceps, needle mounted in a holder or multi-discs dispenser the appropriate antimicrobial disk were placed, within 30 minutes of applying the discs, the plate was inverted and incubated aerobically at 35-37 ℃. After overnight incubation, the growth was examined around the discs and the diameter of each zone of inhibition was measured.
The Following Antibiotics Were Used
Vancomycin (30 mcg), Ampicillin/Cloxacilli (10 mcg), Nitrofurantion (300 mcg), Clindamycin (2 mcg), Cefoperazon (75 mcg), Ertapenam (10 mcg), Aztreonam (30 mcg), Trimethoprim/sulphamethoxazole (1.25/23.75 mcg), Rifampin (5 mcg) and Levofloxacin (5 mcg)
Instruments and Equipment
Bottles, Wear-loop, Incubator, Pasture-pipettes, Refrigerator, Balances, Slides and Coverslips, Micropipettes, Tips for pipettes, Containers, Funnels, Syringes, Pencils & Pen marking, Forceps, Cotton-swab & Gauze, Filter-paper, Beakers Cylinder and Bunsen burner.
Data Analysis
Data entry and Analysis was done using statistical package for social sciences [23]. The quantitative data was analyzed using descriptive statistics. Summarized and displayed on graphs and charts.
Results
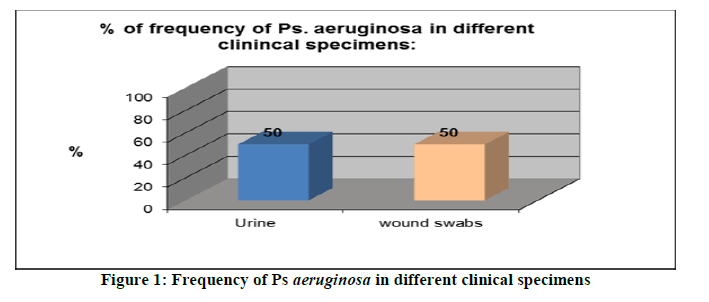
This study included 10 studied bacteriological specimens [Pseudomonas aeruginosa] were who admitted to the main Hodeidah hospital and laboratory Hodeidah city during a period of three months, starting in May 2020 and ending in August 2020. Their bacteriological specimens [Pseudomonas aeruginosa] was taken from patients with wound infection and urinary tract infection, were nearly similar with a percentage of 50% and 50% respectively. The detailed results of this study are presented in the following Table 1 and Figure 1:
| Sample type | Frequency of Ps aeruginosa | |
|---|---|---|
| Number | Percent | |
| Wound swabs | 5 | 50 |
| Urine | 5 | 50 |
| Total | 10 | 100 |
Table 1: Frequency of Ps aeruginosa in different clinical specimens
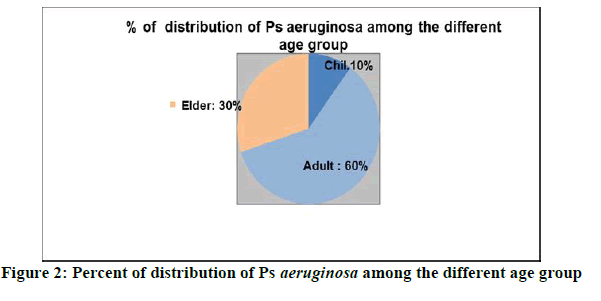
Table 2 and Figure 2 show the 10 studied bacteriological specimens [Pseudomonas aeruginosa] according to their age groups, where the highest age group Adult with a percentage of 60% and the lowest were found in the age group from children with a percentage of 10%.
| Age group | Distribution of Ps aeruginosa | |
|---|---|---|
| Number | Percent | |
| Children | 1 | 10 |
| Adult | 6 | 60 |
| Elder | 3 | 30 |
| Total | 10 | 100 |
Table 2: Distribution of Ps aeruginosa among the different age group
Table 3 and Figure 3 show the antimicrobial resistance patterns of Ps aeruginosa were analyzed for ten different types of antimicrobial agents as mentioned in the method section. We found that Ps aeruginosa was highly resistant to six types are Vancomycin, Ampicillin/cloxacilli, Nitrofurantion, Clindamycin, Cefoperazon and Ertapenam with a percentage of 100%. These results were also not statistically significant.
| Types of antibiotics / No. of specimens | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | No.of resistance |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Aztreonam 50 mcg [AT] | S | I | S | S | I | S | I | S | S | S | 3 |
| Vancomycin 10 mcg[VA] | R | R | R | R | I | R | R | R | R | R | 10 |
| Levofloxacin 5 mcg[LE] | S | R | S | S | I | S | R | S | R | R | 5 |
| Ampicillin\Cloxacilli 10 mcg [AX] | R | R | R | R | R | R | R | R | R | R | 10 |
| Nitrofurantion 200mcg [NIT] | R | R | I | R | R | R | R | R | R | R | 10 |
| Clindamycin 2 mcg [CD] | R | R | R | R | R | R | R | R | R | R | 10 |
| Cefoperazon 75 mcg [CPZ] | I | R | I | I | R | I | I | I | I | I | 10 |
| Ertapenam 10 mcg [ETP] | R | R | R | R | R | R | R | I | R | R | 10 |
| Rifampin 5 mg [RIF] | I | R | R | I | I | I | I | S | I | I | 9 |
| Trimethoprim/sulphamethoxazole[COT] | R | R | R | I | I | S | R | S | R | R | 8 |
Key: S: sensitive, R: resistance, I: intermediate According to BiogramTM any intermediate conceder resistant
Table 3 : Antibiotics-resistance phenotype of all specimens [Ps aeruginosa]
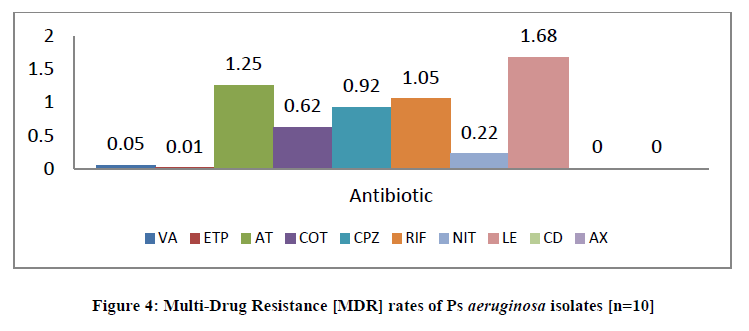
Table 4 and Figure 4 show the mean of different antimicrobial with ten bacteriological specimens [Pseudomonas aeruginosa] Aztreonam and Levofloxacin are sanative to Ps aeruginosa with percentage [20%], while the other resistant with percentage [80%].
| Antibiotic | Expected option | Mean | P |
|---|---|---|---|
| Vancomycin | R | 0.05 | 0.71 |
| Ertapenam | R | 0.01 | 0.8 |
| Aztreonam | S | 1.25 | 0.9 |
| Trimethoprim/sulphamethoxazole | R | 0.62 | 0.7 |
| Cefoperazon | R | 0.92 | 0.63 |
| Rifampin | R | 1.05 | 0.9 |
| Nitrofurantion | R | 0.22 | 0.6 |
| Levofloxacin | S | 1.68 | 0.78 |
| Clindamycin | R | 0 | 0 |
| Ampicillin \cloxacillin | R | 0 | 0 |
Table 4: Multi-Drug Resistance [MDR] rates of Ps aeruginosa isolates [n=10]
Discussion
Ps. aeruginosa present a severe therapeutic challenge and a major health threat globally. Ps. aeruginosa has defined as one of the most common nosocomial pathogens [10]. Hence we have undertaken this study to analyze the antimicrobial susceptibility pattern of Ps. aeruginosa from various clinical samples of a private hospital. Periodic antimicrobial resistance monitoring in Ps. aeruginosa is fundamental to updating the current activity level of commonly used anti-pseudomonal drugs [11]. The present study, 50 % of the Ps. aeruginosa isolates were obtained from wound samples and 50% was isolates from urinary tract infection; Respiratory isolates [42.5%] were the most frequently encountered Ps. aeruginosa isolates from respiratory tract as observed in a similar study of inpatient isolates done in a Saudi Arabian hospital [12].
In the present study, the maximum clinical isolates of Ps. aeruginosa were isolated from adults [60%], followed in Elder [30%] and children [10%].This was similar to study of “Prevalence of Pseudomonas aeruginosa [P. aeruginosa] and Antimicrobial Susceptibility Patterns at A Private Hospital in Sana'a, Yemen [13].
According to the study results measures the rate of resistant of Pseudomonas aeruginosa [n=10,80%] as which is high than previous studies as by Raman et. al. [n=37,67%] [14]. The most important risk factors are obvious, such as excessive consumption of antibiotic exerting pressure on bacteria, the frequent use of invasive devices and relative density of a susceptible patient population with severe underlying disease [15].
In this study, we found that the isolates showed sensitive to Aztreonam [70%] and Levofloxacin [50%]. Our results are in agreement with study carried out by Rashid A.et al. Who documented that Ps. aeruginosa was sensitive to Colistin [86%], Levofloxacin [73%] and Aztreonam [50%] [16]. In our study Ps. aeruginosa showed resistance to Vancomycin [92%], Ampicillin/cloxacilli [100%], Nitrofurantion [100%], Clindamycin [100%] Cefoperazon [100%], Ertapenam[100%] and Rifampin [90%], Al-KabsiA M, et al. found that the resistance rate of Ps. aeruginosa to Ceftazidime [91%], Amikacin [48.4%], Tobramycin [50%] and Piperacillin [91.94%] [17] which is in agreement with our study Seriously, multi-drug resistance of Ps. aeruginosa and other bacteria are consider a problem which must be solve, either by avoiding empirical or random use of antibiotic and treatment should be done using antibiotic susceptibility tests, should be regularly inspected for Ps. aeruginosa colonization which show a strong resistance of Ps. aeruginosa to the various antibiotics.
Conclusion
The conclusion, results of the present study clearly demonstrated the occurrence of resistance to various antipseudomonal agents among the Pseudomonas aeruginosa isolates. The statistics in this study showed high rates of antibiotic resistance to Vancomycin, Ampicillin/cloxacilli, Nitrofurantion, Clindamycin, Cefoperazon and Ertapenam and Rifampin against Pseudomonas aeruginosa strains. We suggest a more restricted and a more rational use of these drugs in hospital setting in order to avoid rapid emergence of resistant strains. Regular anti-microbial susceptibility monitoring is essential of local, regional and national level isolates. This would held and guide the physicians in prescribing the right. Every effort should be made to prevent spread of resistant organisms.
Conflict of Interest
The authors declare that there is no conflict of interests
Acknowledgement
This research was performed with support of the University of Sciences and Technology, Yemen, Hodeida Branch.
References
- PR Murray; EJ Baron; JH Jorgensen; MA Pfaller; RH Yolken. Manual of Clinical Microbiology, American Society of Microbiology, Washington DC, 2003.
- L Collier; A Balows; M Sussman. Topley & Wilson’s Microbiology and Microbial Infections. Arnold London, 1998, 1–6.
- MW Silby; C Winstanley; SA Godfrey; SB Levy; RW Jackson. Pseudomonas genomes: diverse and adaptable. FEMS Microbiol Rev. 2011, 35, 652-680.
- F Barbier; A Andremont; M Wolff; L Bouadma. Recent advances in epidemiology and management. 2013, 216-228.
- Z Pang; R Raudonis; BR Glick; TJ Lin; Z Cheng. Biotechnology Advances. 2019.
- MW Azam; AU Khan. Drug Discov. 2019, 24(1), 350–359.
- RE Hancock; DP Speert. Drug Resist Updat. 2000, 3, 247-255.
- EB Breidenstein; C Fuente-Nunez; RE Hancock. Trends Microbiol. 2011, 19, 419-426.
- LR Mulcahy; JL Burns; S Lory; K Lewis. J Bacteriol. 2010, 192, 6191-6199.
- SY Park; HJ Park; SM Moon; KH Park; YP Chong; SH Choi; et al. BMC Infect Dis. 2012, 12, 308.
- EB Hirsch; VH Tam. Expert Rev Pharmacoecon Outcomes Res, 2010, 10, 441-451
- H Hanan. International J infectious diseases. 2006, 10(4), 326-333.
- A Alyahawi; AM Alhomidi; NA Henhena. J Pharmaceutical Research. 2018, 3(4), 12-15.
- R Gowri; E Avendano; J Chan; S Merchant; L Puzniak. Antimicrobial Resistance & Infection Control. 2018, 7(1), 79.
- A Rashid; A Chowdhury; SHZ Rahman; SA Begum; N Muazzam. J Med Microbiol. 2007, 1(2), 48-51.
- M Dash; S Padhi; MV Narasimham; S Pattnaik. J Health Sci. 2014, 3, 15-19.
- M Dash; S Padhi; MV Narasimham; S Pattnaik. J Health Sci. 2014, 3, 15-19.